The earliest recorded attempts at hip replacement date back to 1891. At the time, ivory was proposed to replace the femoral heads of patients whose hip joints had been destroyed by tuberculosis. Since then, everything changed. THINK speaks to the MaltaHip Team to learn about their innovative hip replacement technology.
Continue readingLiving with a rare disease
Author: Clayton Axiak

Picture yourself waking up one morning with a severe, relentless itch that no clinician or diagnostic tool can understand. Your life would be thrown off kilter. Quality of life would suffer financially, psychologically, and socially as you try to look for a glimmer of light at the end of the tunnel. This is what life is like for most people living with a rare disease.
Often barraged with terms like ‘unknown’ or ‘undiagnosed’, matters can get even more challenging when the condition becomes more elusive or develops life-threatening consequences. And all of this is exacerbated by inequities in treatment and high costs of the few existing drugs that are available.
By EU standards, a rare disease is one that affects fewer than one in 2,000 individuals. And these ‘less common’ ailments are difficult to raise monies for to research, leaving large gaps in scientific and medical literature. One such disease is the poorly understood Idiopathic Hypogonadotropic Hypogonadism (IHH).
Characterised by the absence of puberty and infertility, IHH can be compounded by potentially severe characteristics such as congenital heart disease, osteoporosis at a young age, and early onset of Alzheimer’s disease.
Its cause is usually a genetic anomaly, but a single genetic change can affect two people very differently. This gives rise to an unparalleled complexity that makes the cause harder to decipher. Symptoms are not clear-cut and sometimes mask the actual underlying cause, bringing about misdiagnosis and delayed treatment. Timely diagnosis is crucial for successful treatment that enables the patient to achieve puberty and induce fertility. But this is not always possible.
Under the guidance of Dr Rosienne Farrugia, I am currently analysing and expanding upon a preliminary assessment of IHH in Malta using high-throughput sequencing (HTS) technology (conducted by Adrian Pleven). With HTS, we can read a person’s entire DNA sequence and attempt to identify differences in the DNA code which lead to such diseases.
What the team has found is that some genetic variants typical of IHH are more common in the Maltese population when compared to mainland Europe and African populations. This is likely due to the reduced genetic variation of our population, shaped by successive events of population reduction and expansion throughout our history.
By mapping the genetic cause of diseases prevalent on our islands, we can help medical consultants to employ specific screening tests that are tailored for local patients suffering from IHH. Such advancements in genomic technology and personalised medicine can make a huge impact on people’s lives. And not only to those suffering from IHH; researching one disease, however rare it may be, can shed light on mechanisms that prove useful in treating many others, ensuring that when it comes to health, no one is left behind.
This research project is being carried out as part of a Ph.D. program in Applied Biomedical Sciences at the Faculty of Health Sciences.
Diabetes: from genes to blood

Type 2 diabetes mellitus is a disease that affects over 250 million people worldwide. Many in Malta suffer from the disease because of our high carbohydrate diet and lack of physical activity. Type 2 diabetes arises when levels of the sugar glucose remain very high in the blood. Testing normally involves frequent finger pricks to determine blood sugar levels, or otherwise a patient can take a sugary drink followed by regular urine/blood testing over 2 or more hours.
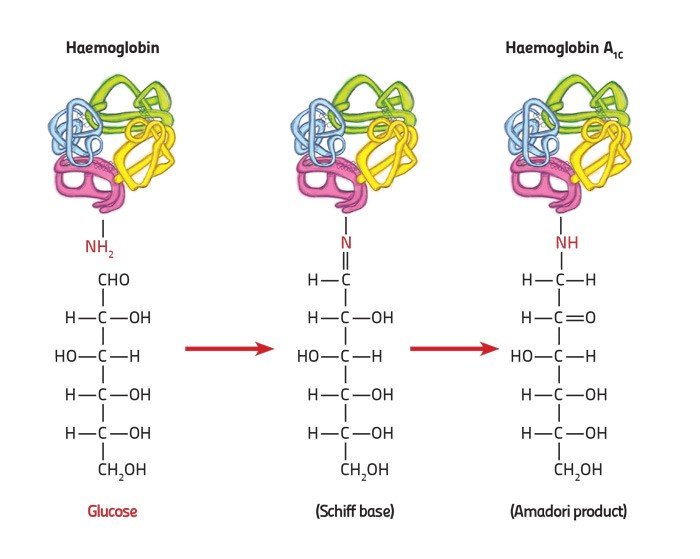
Alexandra Fiott (supervised by Prof. A. Felice) studied whether the absolute HbA1c levels (the haemoglobin fraction with sugar attached multiplied by the haemoglobin concentration) would provide a better method to describe the link between one’s genetics and diabetic condition. She attempted to reduce the frequency of the testing needed while using a relatively non-invasive test — the withdrawing of one tube of blood, while investigating the genetics of diabetes.
Haemoglobin (Hb) transports oxygen throughout the blood through red blood cells. The HbA1c forms when glucose binds to haemoglobin. This can be used as an indirect measure of average blood sugar concentrations. Measuring HbA1c levels is rapid, but unfortunately the results are influenced by factors that affect red blood cells. With around 5% of Maltese having red blood cell disorders, an alternative measurement would help reduce inaccurate results and unnecessary worry for patients. The absolute HbA1c was used for this study.
The genetics and blood profile of five different patient groups were determined using genetic and biochemical methods: adults with a normal blood profile, anaemics, beta-thalassaemics, pregnant women, and type 2 diabetics (on limited treatment). Statistical analysis did not reveal an improved link, but the absolute HbA1c did help distinguish between the different patient groups.
To improve the reliability of these results, a separate set of experiments was carried out to see whether a known Maltese variation in haemoglobin, with a prevalence of around 1.8% in the Maltese population, has an effect on the amount of sugar that binds to the haemoglobin. This variant was found not to influence the blood glucose levels and therefore the HbA1c.
Taken together these results showed that the absolute HbA1c does not improve the link between the genetics and blood profile of the patients. However, it could distinguish between different groups of patients.
This research was performed as part of an M.Sc. (Melit.) in Biomedical Sciences at the Faculty of Medicine and Surgery at the University of Malta.