COVID-19 vaccines are very good at equipping our bodies with immune defences required to fight the virus should we get infected. However, immunocompromised people may not be protected even if they have received all doses of a vaccine. An international project aims to engineer antibodies that tackle the virus. Bacteriophages can be an important tool to develop these.
Infectious diseases are generally treated by strengthening the body’s defensive mechanisms so it can ward off the attackers and weaken or destroy them. When COVID-19 struck, scientists across the world began the race to find adequate treatments for the disease. They explored the potential of using existing drugs as treatment options and also developed new vaccines.
COVID-19 vaccines train the body’s immune system to readily identify SARS-CoV-2 and attack it. Thus, they reduce the severity of infections if and when they occur. While the world heaved a sigh of relief for the development, approval, and administration of these vaccines, some people may still be unprotected against the virus despite being up-to-date with vaccine boosters because they are immunocompromised. That is, their bodies’ immune systems are not able to respond to health threats as they should due to diseases, treatments, age, or other reasons.
Immunocompromised patients may not produce protective antibodies against the virus in response to infection or vaccination. Hence, they remain at risk of severe symptoms when infected with SARS-CoV-2. Scientists are seeking options to help the immunocompromised fight COVID-19, as well as other diseases.
Bacteria Killers to the Rescue
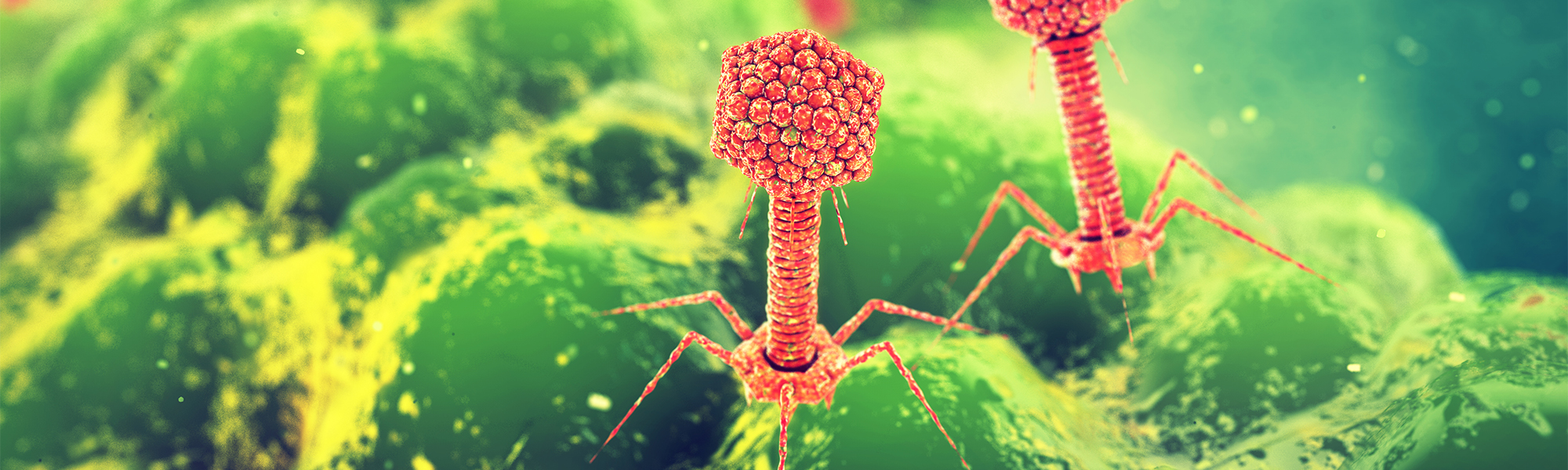
One prominent option is the use of bacteriophages, or ‘phages’, a type of virus that infects bacteria.
‘Phages are like molecular machines that specialise in killing bacteria,’ says Prof. David Saliba, Associate Professor at the University of Malta’s Department of Applied Biomedical Science. Prof. Saliba and his team are working to produce antibodies against SARS-CoV-2 and Zika viruses. They are investigating how phages can be used to identify antibodies that target these viruses. But how do bacteriophages operate?
In nature, phages are efficient predators that have intricately engineered needles that can bind to different parts of bacteria. They then inject the bacteria with their genetic materials. Once inside, phages hijack the bacterial cell’s biological processes and produce identical copies of themselves, destroying their host in the process. Phages are specific in regards to the bacteria they infect. That is, a particular phage can only infect particular species of bacteria. Within a lab, phages have been modified into a tool for molecular biology. Building upon the work of 2018 Nobel Prize in Chemistry winners George P. Smith and Sir Gregory P. Winter, scientists are now able to manipulate phages to screen for therapeutic agents, such as antibodies, allowing for the development of new treatments.
Phage Display
Antibodies stop infections by attaching themselves to invading pathogens. They bind to the parts that pathogens use to infect human cells.
One characteristic of phages is that they exhibit (display) some products of their genetic material on their surfaces. Phages can be modified to display either antibodies, small protein fragments (peptides), or other biomolecules on their surface. Through a collaboration with the University of Edinburgh and the International Centre for Cancer Vaccine Science in Gdansk, the team has a collection of over a billion different antibodies displayed on phages at their disposal. This collection of antibody-displaying phages is called a ‘phage library’, with each individual antibody having a specific target to which it can bind. Scientists in Prof. Saliba’s lab are sifting through this collection to identify individual antibodies that specifically target the SARS-CoV-2 and Zika viruses. Once purified, these individual antibodies could be used prophylactically (that is: to prevent the spread or occurrence of a disease or infection) as well as therapeutically by immunocompromised patients.
Since antibodies bind specifically to their targets, the team has to identify phage-displayed antibodies that bind to a dud virus which cannot replicate.
‘We created 3-dimensional magnetic beads coated with SARS-CoV-2’s spike protein,’ says Giuseppina Monda, a research scientist on the team. ‘We expose them to the antibody phage library so that only the phages that display SARS-CoV-2-specific antibodies can bind to them,’ she continues.
The team then collects and amplifies the antibody phages specific to the spike protein of SARS-CoV-2 and repeats the process a few times over to further isolate a pure collection of virus-specific antibodies. The process is referred to as ‘biopanning’ as it is akin to the process used to find gold from a riverbed.
The next step is to test the neutralising ability of the antibodies to determine whether they have the potential to stop the virus from infecting human cells. The team has produced phage antibodies against the Zika dud virus and is conducting similar neutralising tests on them.
With these, the antibodies produced by the team will not only weaken the virus, but stop the virus in its tracks. Thus, immunocompromised persons can have a reliable form of protection against diseases such as SARS-CoV-2 and the Zika virus.
Stay tuned for the March 2023 edition of THINK, where we will share results from the project!
Project ACCELERATE is funded by the Malta Council for Science and Technology for and on behalf of the Foundation for Science and Technology, through the Infectious Disease Programme.




Comments are closed for this article!